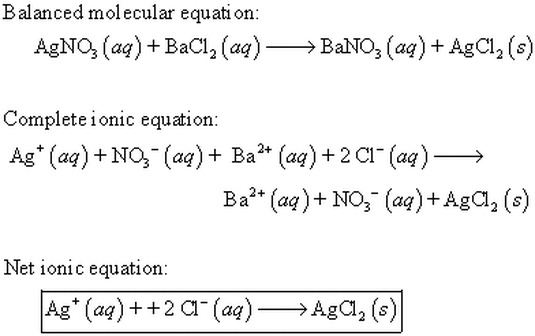1. Periodic Table and Elements Review
2. Nomenclature Review
3. Types of Chemical Reactions
|
If we are able to recognize the type of reaction we will be able to predict the products that are formed.
|
|
1. Synthesis Reactions
|
|
Two or more simple substances combine to form a more complex compound.
Formation of Magnesium Oxide |
2. Decomposition Reactions
|
|
A compound breaks down into simpler substances
|
3. Combustion
|
|
These reactions usually give off large amounts of energy in the form of heat and light.
Some involve Hydrocarbons and will always produce carbon dioxide and water. As well, anything reacting with Oxygen (other than Hydrogen) is considered a combustion reaction |
4. Single Displacement Reaction
|
|
Occurs when one element is replaced by another in a compound.
Also called Single Replacement reactions |
5. Double Displacement Reaction
|
|
Occurs when elements in the reacting compounds switch places with each other
|
6. Neutralization Reactions
|
|
Occurs when an acid reacts with a base
The products are always a salt and water. Remember salt is an ionic compound. |
4. Ionic Equations
|
|
Dissociation
When ionic compounds dissolve to produce ions the process is typically called dissociation. Dissociation of ionic compounds occurs when water molecules “pull apart” the ionic crystal. This occurs due to strong attractions between the polar ends of the water molecule and the positive and negative ions within the crystal. Water molecules then surround the positive cations and negative anions; this is called hydration (https://www.spiritsd.ca/curr_content/chem30_05/5_acids_bases/acids1_3.htm) |
|
Double Displacement Reaction where a precipitate (solid) forms
|
5. The Mole
|
|
The word “mole” suggests a small, furry burrowing animal to many. But in this lesson, we look at the concept of the mole in chemistry. Learn the incredible magnitude of the mole--and how something so big can help us calculate the tiniest particles in the world
|